SOLVED: A 25 gram piece of hot metal at 97°C is plunged into a 35 gram cup of cool water at 19°C. The metal gives up its heat to the water until
5 (300) · $ 25.00 · In stock
VIDEO ANSWER: in this question, there is a cup in this cup there is a water. The specific head of the water is given us 4.18 June program, degree seven degree. The water is at 19°C. The 25 g Piece of Hot Metal. This is very hot metal. The temperature
Numerade is a venture-backed, high-growth education technology startup based in Pasadena. We are singularly focused on creating exceptional video and interactive content experiences for education making the knowledge and skills of world class educators widely accessible and affordable to student audiences of all backgrounds. Our mission is to close the educational opportunity gap by unlocking and democratizing access to extraordinary educators and the content they have to offer.

Answered: Question 8 A 25 g sample of metal is…

A 155.0 g piece of copper at 168 oc is dropped into 250.0 g of water at 20.9 oc. (the specific heat of
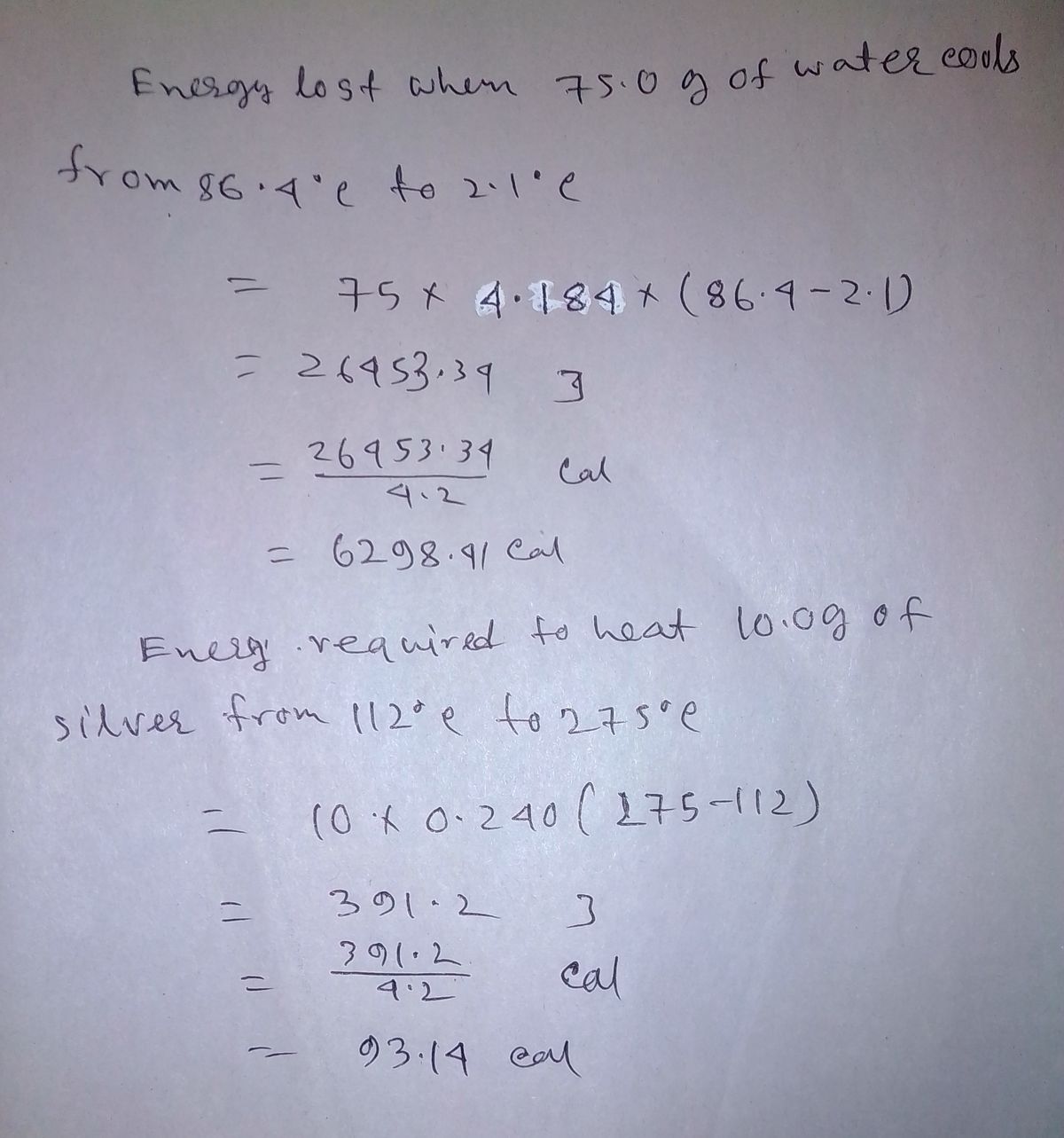
Answered: Answer in joules and calories. Energy…

SOLVED: A 25 gram piece of hot metal at 97°C is plunged into a 35 gram cup of cool water at 19°C. The metal gives up its heat to the water until

Measurement of discharge as inflow into leaky reservoirs

2021 in science - Wikipedia

2001 VIDEO CATALOG - Clinton Essex Franklin Library System

Liquid Drain Clog Dissolver - 31oz

Suppose that 0.50 g of water at 25 ∘c condenses on the surface of a 51-g block of aluminum that is

Solved 3. A 32.520 g piece of unknown metal was heated in a

Spain - Wikipedia

5.22 A 70.0-g piece of metal at 80.0 °C is placed in 100 g of water at 22.0 °C contained in a

Specific Heat Pre – Lab. Specific Heat Different substances require different amounts of heat to change their temperature. In general the specific heat. - ppt download

14.2 Temperature Change and Heat Capacity





