CORE-A End of Therapy (EoT) information : Clinical Outcomes in
4.9 (320) · $ 29.99 · In stock
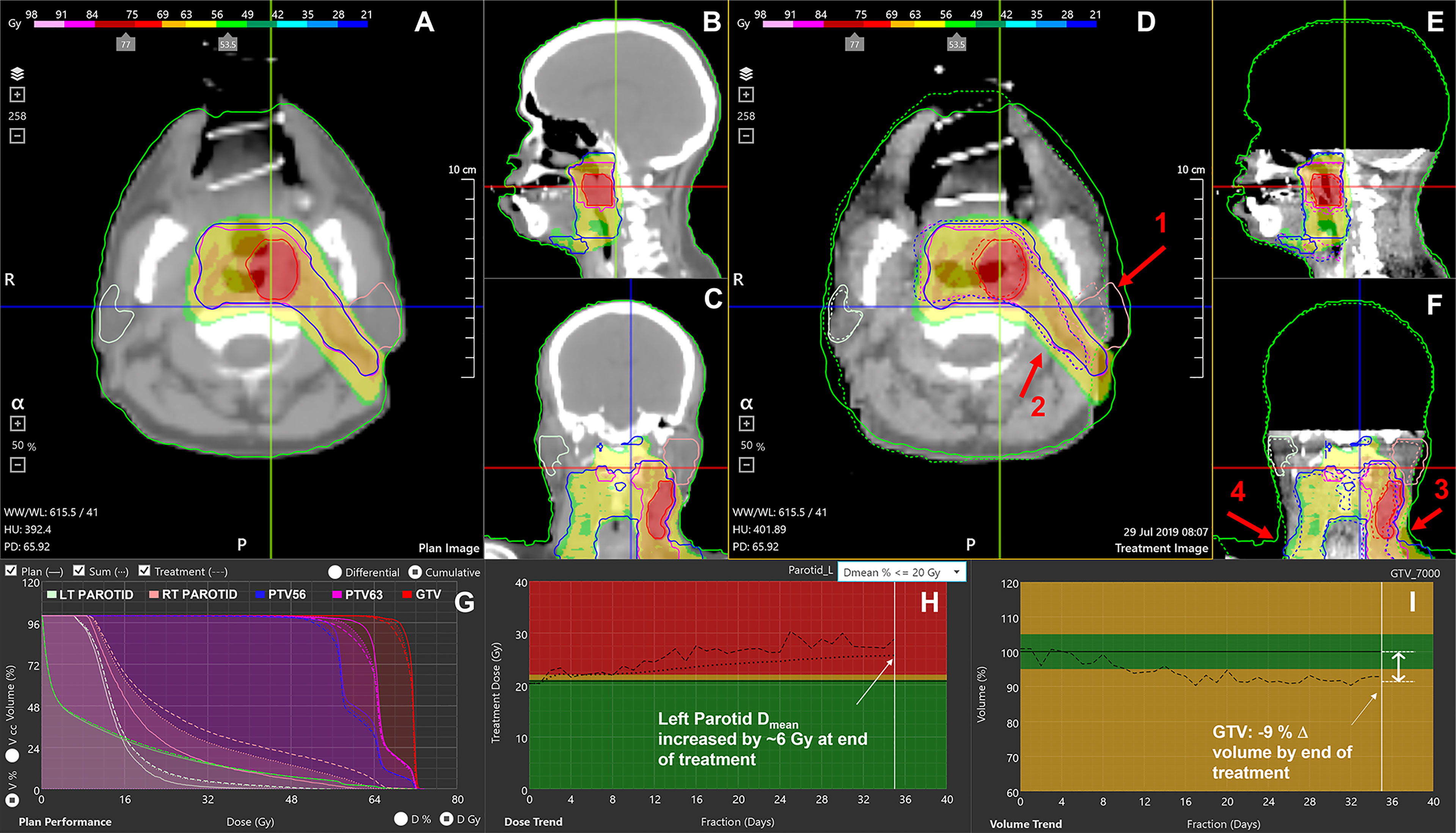
Frontiers Retrospective Clinical Evaluation of a Decision-Support Software for Adaptive Radiotherapy of Head and Neck Cancer Patients
Clinical Outcomes in Routine Evaluation (CORE-OM) – NovoPsych
CORE-A : Clinical Outcomes in Routine Evaluation (and CST)

Aspects of the CORE system Chris Evans 5 th Conference on Psychiatric Research in the North, Stokmarknes, Norway, 3.ix.03. - ppt download

Change from baseline in EORTC QLQ-C30 pain scores by treatment arm. C
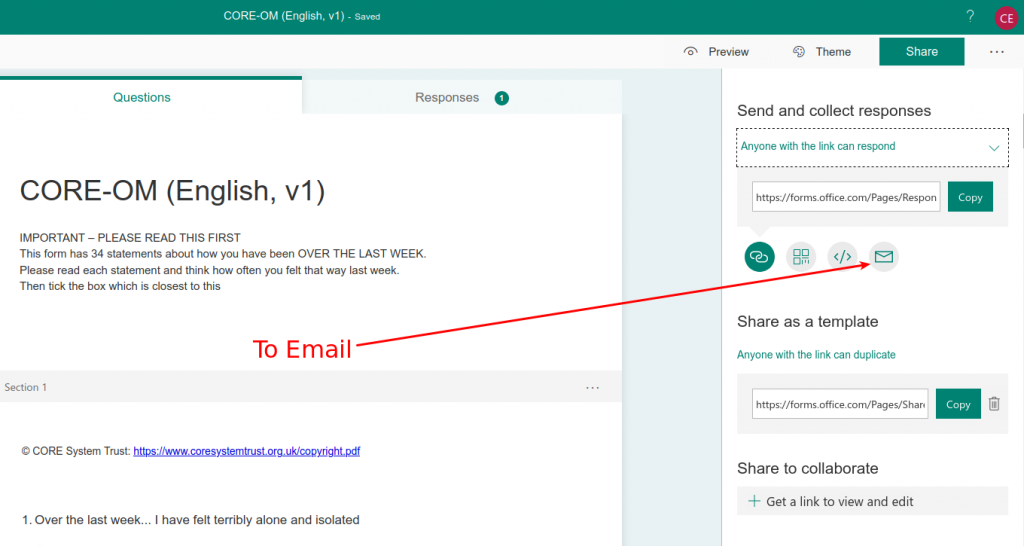
Microsoft forms for CORE measures : Clinical Outcomes in Routine Evaluation (and CST)

SF-36 outcomes n = 17 Baseline EOT ∆ EOT -Baseline
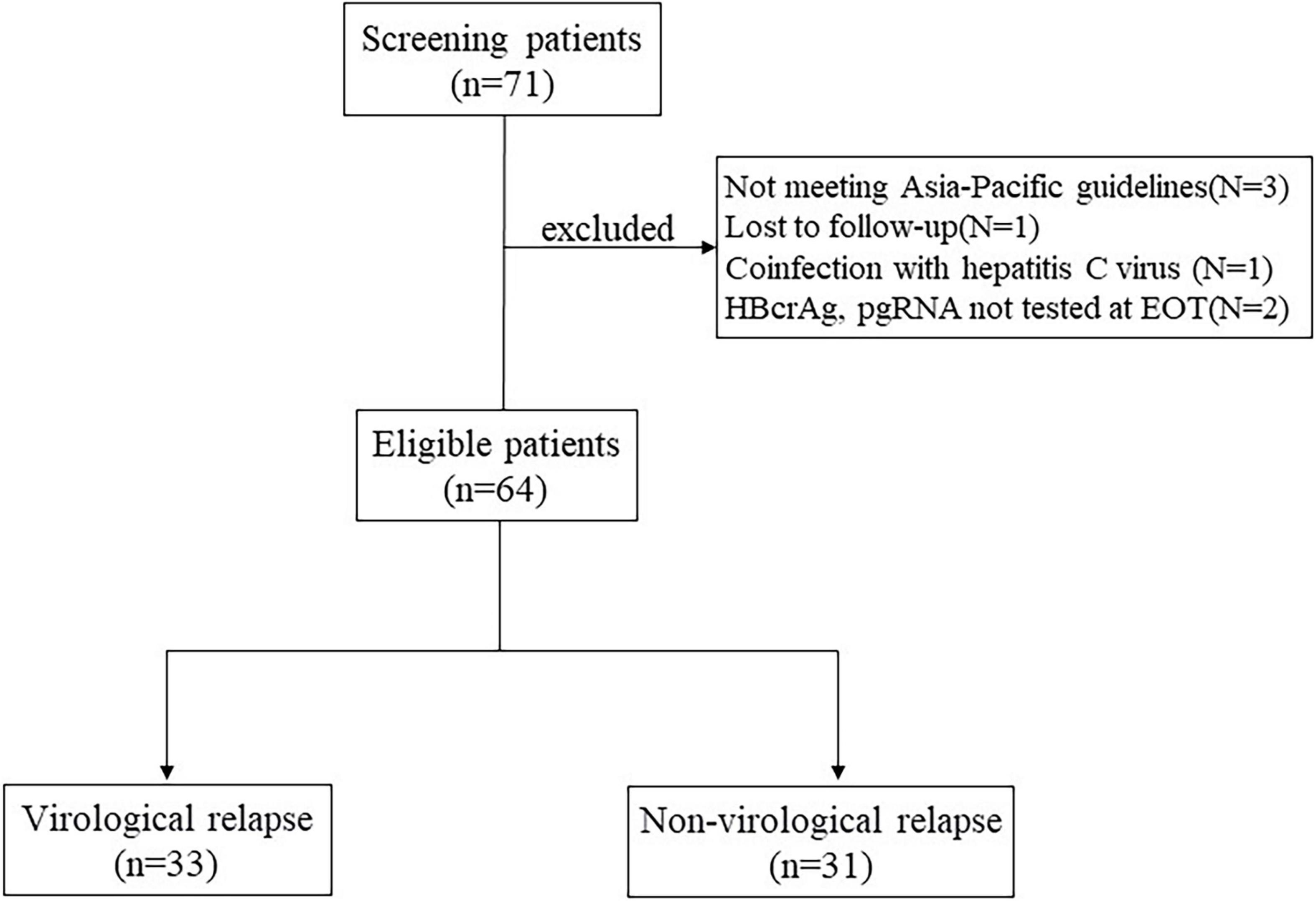
Frontiers Serum Pregenomic RNA Combined With Hepatitis B Core-Related Antigen Helps Predict the Risk of Virological Relapse After Discontinuation of Nucleos(t)ide Analogs in Patients With Chronic Hepatitis B
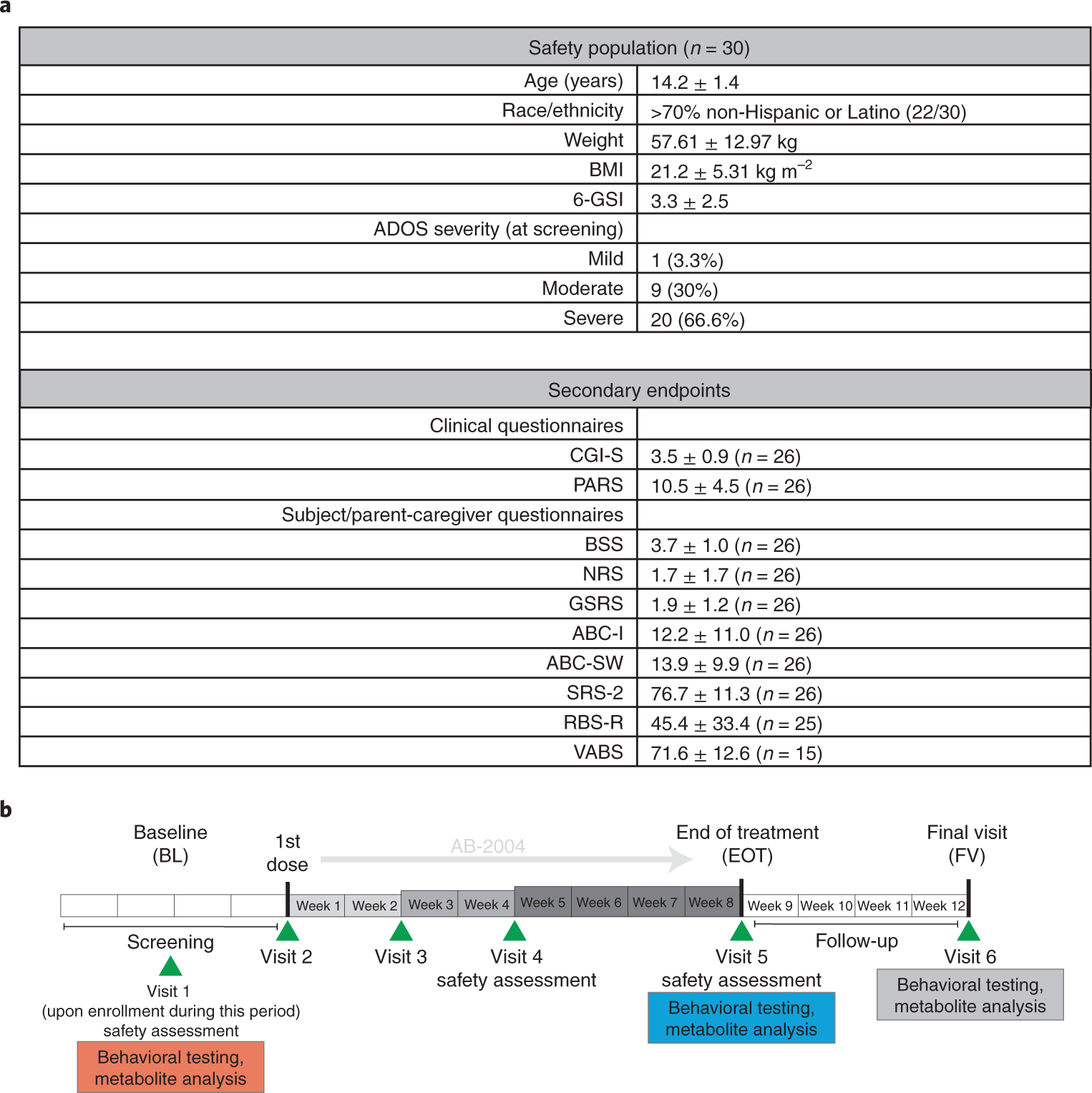
Safety and target engagement of an oral small-molecule sequestrant in adolescents with autism spectrum disorder: an open-label phase 1b/2a trial
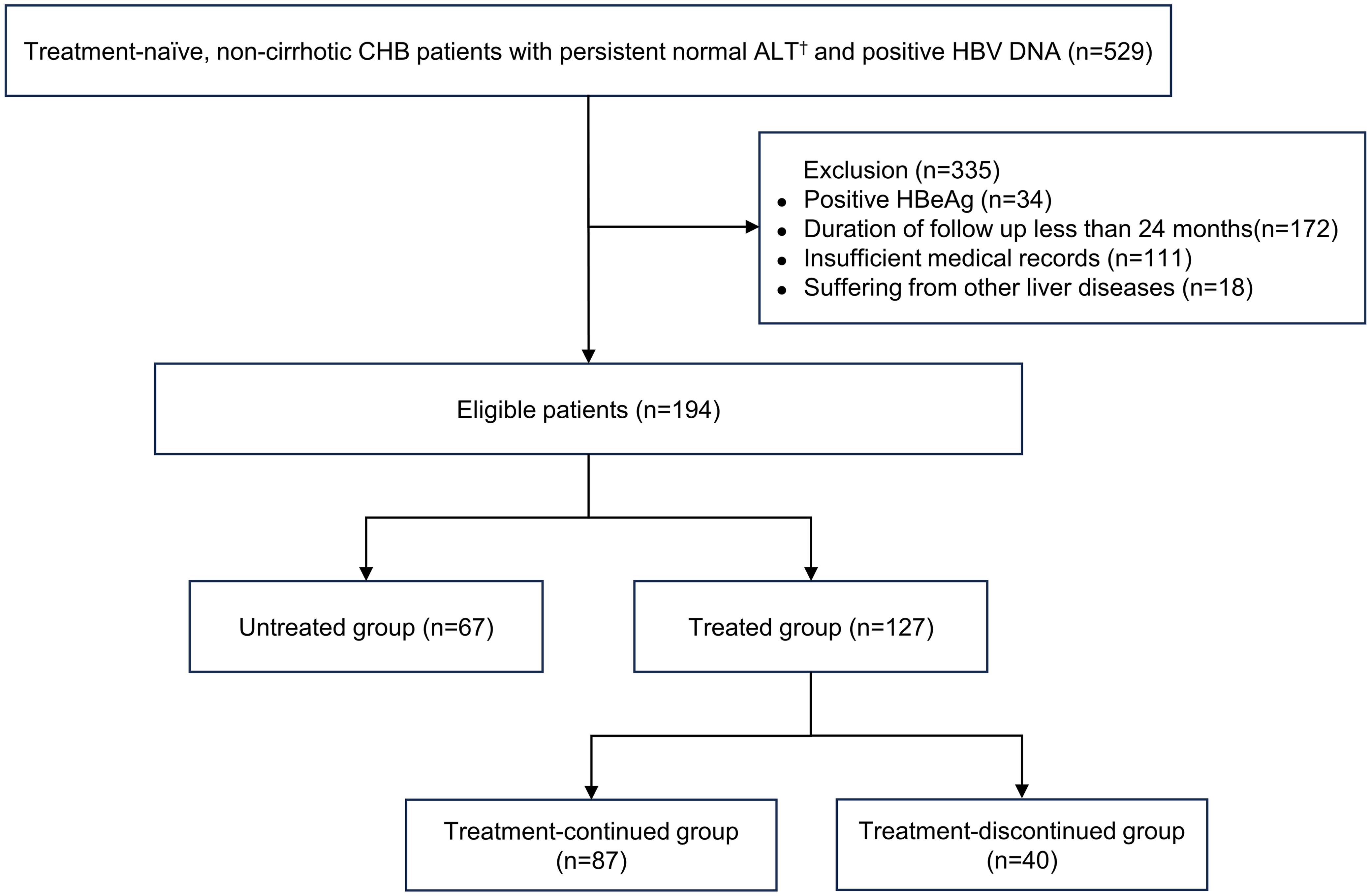
Antiviral Therapy Favors a Lower Risk of Liver Cirrhosis in HBeAg-negative Chronic Hepatitis B with Normal Alanine Transaminase and HBV DNA Positivity
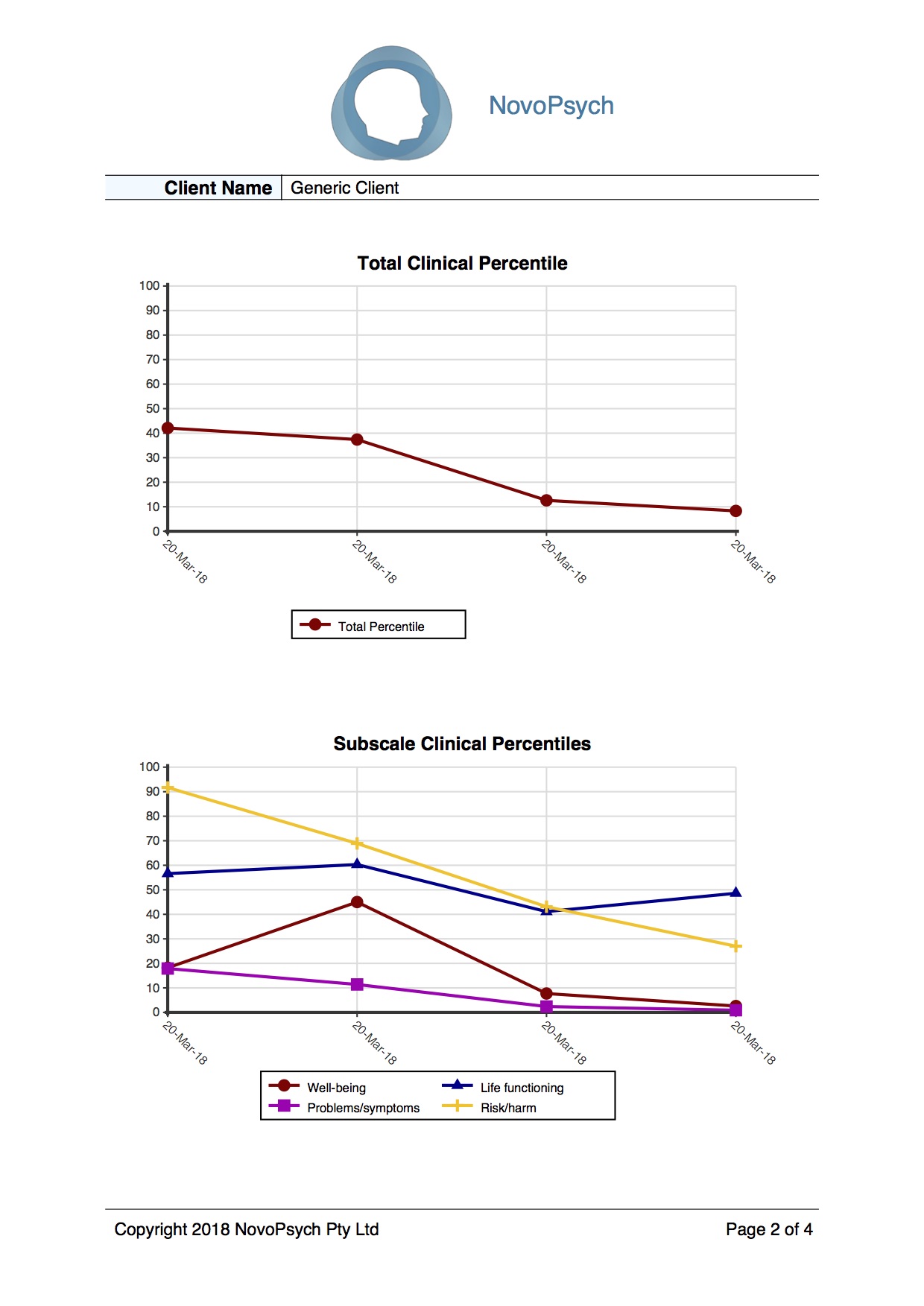
Clinical Outcomes in Routine Evaluation (CORE-OM) – NovoPsych







